Vaccination Requirements
Images are not of actual patients
Meningococcal Vaccination Recommendations
Risk Evaluation and Mitigation Strategy (REMS) program
Due to the risk of serious meningococcal infections, ULTOMIRIS® (ravulizumab-cwvz) is available only through a restricted program under a REMS. Enrollment in the REMS program and additional information on the requirements are available at UltSolREMS.com or by calling 1-888-765-4747.1
WATCH: A gMG Expert Reviews Vaccination Requirements and Alexion’s REMS Program
In this video, Dr. Jon Durrani provides an overview of Alexion's REMS program and the needed vaccinations for ULTOMIRIS.
Explore real-world data from over 14 years of post-marketing experience for ULTOMIRIS and SOLIRIS
Download MeningococcalInfection Rates Brochure
Advisory Committee on Immunization Practices (ACIP) guidelines
Complete or update meningococcal vaccination (for serogroups A, C, W, Y and B) at least 2 weeks prior to administration of the first dose of ULTOMIRIS, per the current ACIP recommendations for patients receiving a complement inhibitor.1
- ACIP recommends that persons using complement inhibitors should complete or update vaccination at least 2 weeks before complement inhibitor initiation unless the risks of delaying treatment outweigh the risks of developing meningococcal disease2
- Revaccinate patients in accordance with ACIP recommendations considering the duration of ULTOMIRIS therapy1,a
If patients have not been vaccinated and ULTOMIRIS must be started right away, provide antibacterial drug prophylaxis and administer meningococcal vaccines as soon as possible.1,b
The benefits and risks of antibiotic prophylaxis for prevention of meningococcal infections in patients receiving ULTOMIRIS have not been established.1
Required Meningococcal Vaccination Regimen
Your patient must receive both MenACWY and MenB vaccine series. The vaccines may be administered during the same visit but at different injection sites.2,3
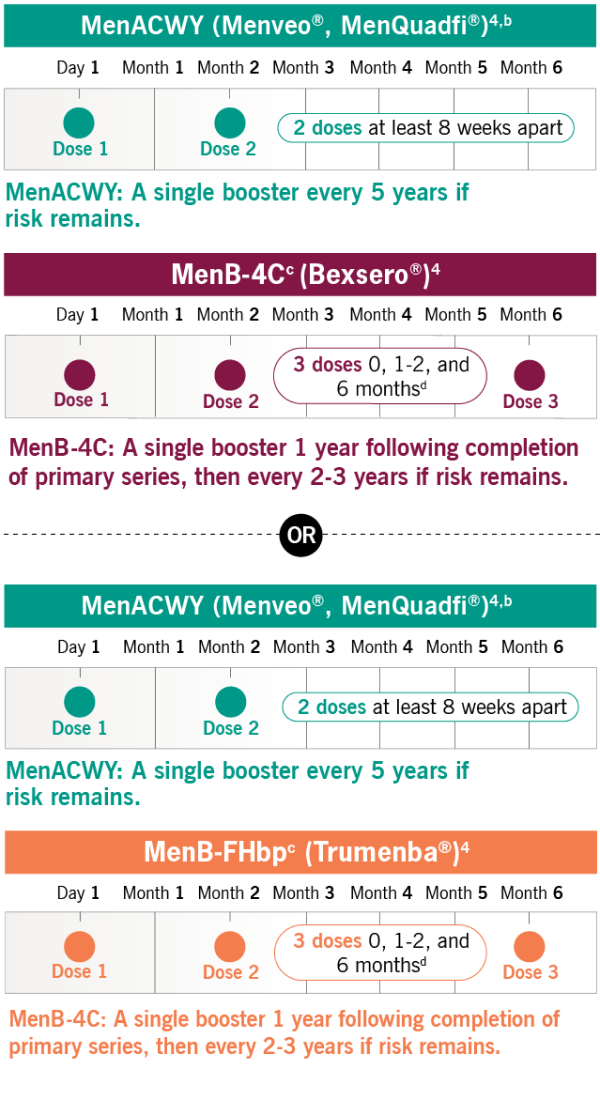
This list is not exhaustive and is intended to provide an example of most commonly prescribed meningococcal vaccines. The choice of vaccine brand deemed medically appropriate is the decision of the treating HCP.
Please see the respective meningococcal vaccine’s Prescribing Information for complete details, including the vaccine’s Warnings, Precautions, and Contraindications.
- If your patient received meningococcal vaccines in the past, they might need additional vaccination before starting ULTOMIRIS3
- The choice of vaccine deemed medically appropriate is your independent decision
- In most cases, your patients can receive meningococcal vaccines at a physician’s office or retail pharmacy
- To help reduce the risk of meningococcal infections, the complete series for the MenACWY and MenB vaccines should be administered3
aNote that ACIP recommends an administration schedule in patients receiving complement inhibitors that differs from the administration schedule in the vaccine prescribing information.1
bSeveral antibiotics are available for the treatment of meningococcal disease, including ceftriaxone, cefotaxime, and, when the diagnosis is confirmed, penicillin.5
cMenB vaccines are not interchangeable. Patients must receive the same product for all doses.4
dFor additional information on clinical considerations, refer to the most current ACIP recommendations and CDC immunization schedule.
